Health officials plan major research on COVID-19 long-haulers
NIH expects studies to be underway by the summer
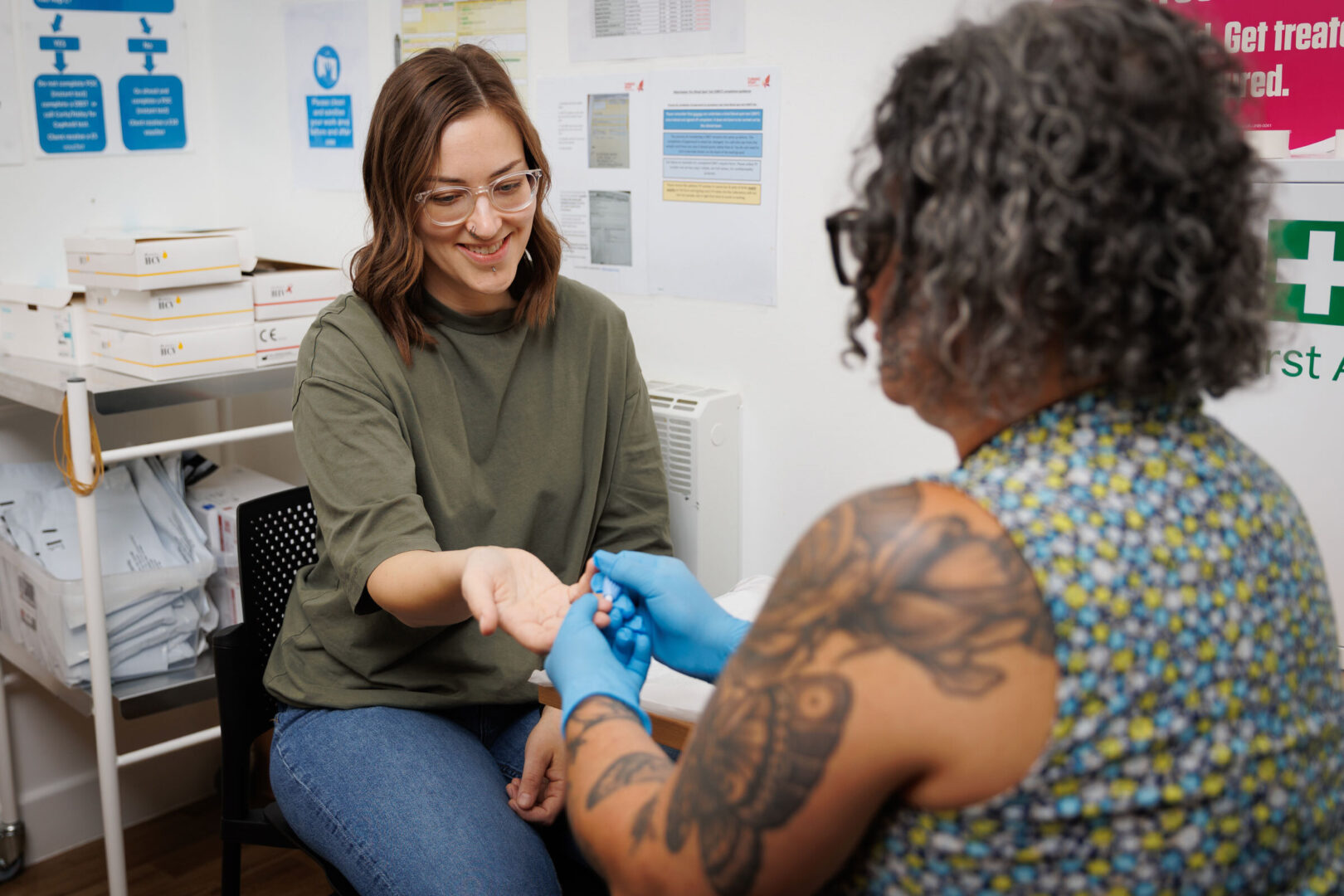
The National Institutes of Health is preparing to award grants in the next three weeks to researchers studying the long-term effects of COVID-19 and patients experiencing “long COVID.”
NIH Director Francis Collins told the Energy and Commerce Health Subcommittee this week that the agency expects laboratory research and imaging studies to be underway by the summer. The agency received 273 research proposals after Congress provided more than $1 billion for research into the long-term effects of COVID-19.
Many people who have been sick with COVID-19, including some who had mild or no symptoms, reported dealing with additional symptoms long after their acute illness ended. The long-haul COVID-19 symptoms can range from fatigue or headaches to mental health issues or chronic pain.
“Some of you have been suffering for more than a year with no answers, no treatment options, not even a forecast of what your future may hold,” Collins said during his opening statement Wednesday, referring to patients suffering from long COVID-19. “Some of you have even faced skepticism about whether your symptoms are real. I want to assure you that we at NIH hear you and believe you.”
Some lawmakers said they’d heard from constituents or even family members dealing with long COVID-19.
The NIH, working with other federal health agencies, is launching a meta cohort of different studies to learn more about the long-term effects and illnesses linked to COVID-19. The research will look at issues such as whether receiving a COVID-19 treatment affects whether a patient may experience long-term effects, what predisposing factors may lead to longer illnesses, and how long COVID-19 may affect children.
Collins said the research should draw from tens of thousands of patients. Participants should reflect diverse backgrounds and a range of COVID-19 symptoms, he added.
[jwp-video n=”1″]
The research, which Congress funded late last year, will draw from electronic health records for patients at major health systems and involvement from patient advocacy groups.
“That $1.15 billion that Congress provided in December for us to build this very intense large-scale study is what we need right now,” said Collins, responding to questions from Rep. Debbie Dingell, D-Mich. “I don’t know exactly what we’re going to encounter as this goes forward. We’re going to want to institute as many clinical trials as possible for interventions. We may need to find more resources.”
Officials are also interested in learning more about anecdotal reports from some patients suffering from long COVID-19 who said they began to see improvements after being vaccinated. Collins told Rep. Brett Guthrie, R-Ky., that it’s been difficult to collect enough data to go beyond anecdotal reports.
“Does that say that there is still lingering infection of this SARS-CoV2 virus in people with long COVID, even though we have not been able to recover the virus from those folks? Does the vaccine getting this response say that there is some sort of reservoir there? It’s one of the more interesting things that’s come along recently,” Collins said.
John Brooks, the Centers for Disease Control and Prevention’s COVID-19 chief medical officer, told the subcommittee that the agency has determined three general types of conditions that patients may develop after having COVID-19.
The first, long COVID-19, includes a range of symptoms that can last for months. Another can affect one or more body systems or an organ, while the third covers complications from prolonged treatment or hospitalization for the virus.
Brooks said the agency is also committed to ensuring health equity in addressing post-COVID-19 illnesses, particularly given that racial and ethnic minorities are more likely to get COVID-19 and less likely to access health care services.
The agency is also following cohorts of patients for up to two years to determine the proportion of patients who develop conditions after being sick and to assess what risk factors may influence whether someone experiences long COVID-19, he said. CDC is also conducting online surveys and reviewing de-personalized electronic health records to gather more information.
Brooks said the agency is also working to develop case definitions related to long COVID-19 to help physicians. He told Rep. Peter Welch, D-Vt., that officials are using machine learning and data sets to determine what sorts of visits patients are making to doctors and what drugs they may be prescribed. The CDC is also interviewing doctors about their patients.
“What really makes this a challenge is the broad heterogeneity, and so we’ve got a lot of work to do,” Brooks said.